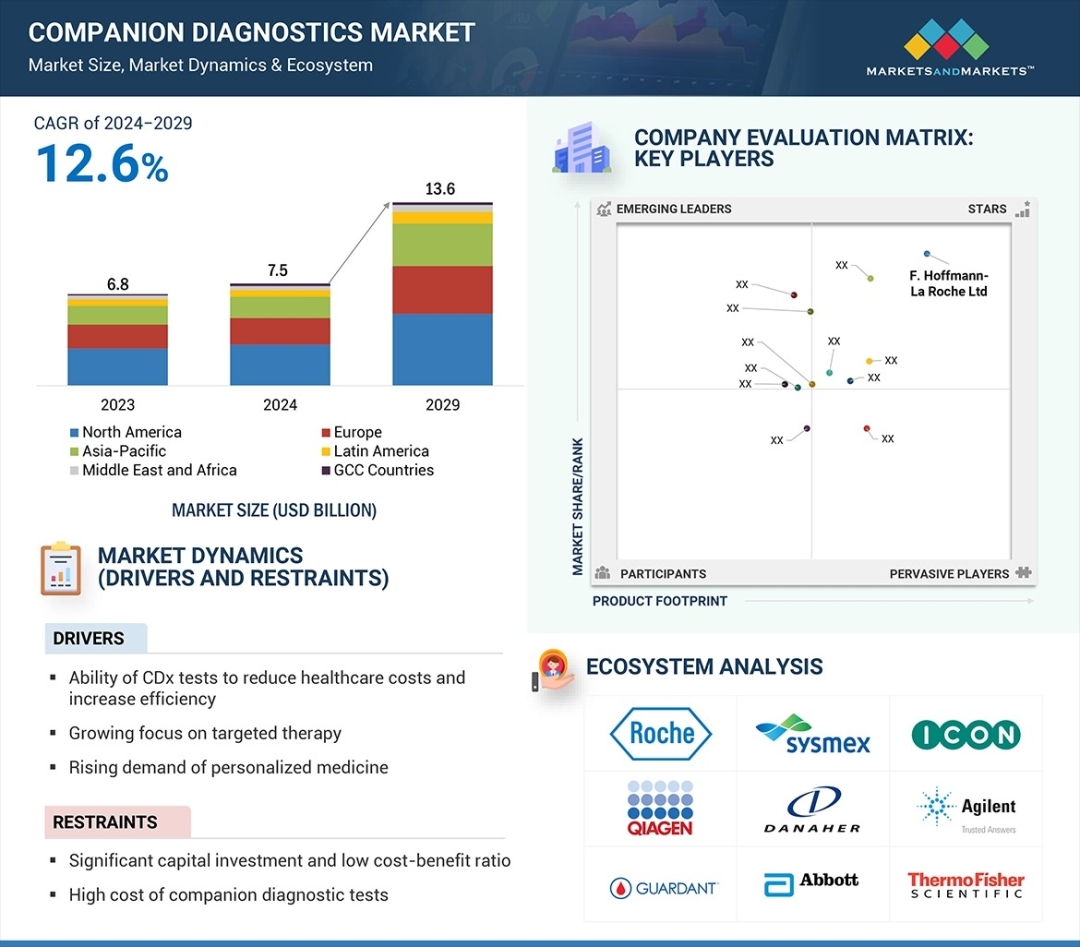
The global companion diagnostics market has entered a transformative growth phase, valued at $7.5 billion in 2024 and positioned to achieve $13.6 billion by 2029, representing a compound annual growth rate (CAGR) of 12.6% over the forecast period. This expansion reflects a fundamental shift in how healthcare systems approach drug development, patient stratification, and therapeutic decision-making at the enterprise level.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=155571681
WHAT IS DRIVING THIS MARKET INFLECTION?
The companion diagnostics sector is experiencing unprecedented momentum driven by three converging forces. First, rapid advancements in medical technology have fundamentally transformed therapeutic approaches, enabling more precise and efficacious treatments. Second, the rising prevalence of cancer globally has intensified demand for targeted therapies that identify patient populations most likely to benefit from specific drug candidates. Third, and most critically, the strategic importance of companion diagnostics in the drug development lifecycle has become non-negotiable for pharmaceutical and diagnostic manufacturers competing for market share.
The market's resilience stems from a deeper structural shift: the healthcare industry's transition toward personalized medicine. Growing clinical awareness of precision medicine's advantages is driving adoption of companion diagnostic and genomics technologies at scale. These diagnostic tests—which analyze specific biomarkers and genetic indications for particular therapeutic interventions—have become integral to clinical decision-making for chronic disease management and oncology, empowering healthcare providers to deliver tailored treatment plans that reduce adverse events and improve patient outcomes.
WHY NOW? THE STRATEGIC IMPERATIVE FOR C-SUITE LEADERS
For chief executives and financial decision-makers, companion diagnostics represent a critical infrastructure investment. By enabling informed, data-driven treatment selections, these tools reduce clinical uncertainty, optimize drug efficacy, and strengthen the value proposition of high-priced specialist therapies. This alignment between diagnostic precision and therapeutic safety directly impacts operational efficiency, regulatory compliance, and competitive differentiation in an increasingly value-based care environment.
HOW IS THE MARKET STRUCTURED? KEY SEGMENTS AND TECHNOLOGY LEADERS
By Product & Service: The assays, kits & reagents segment commanded the largest market share in 2023, reflecting the wide variety of available diagnostic products and their expanding utilization across multiple therapeutic domains. This dominance underscores the market's maturity in core diagnostic offerings while signaling sustained demand for consumable-based solutions.
By Technology: Polymerase chain reaction (PCR) technology maintains the highest market share, driven by its ease of use, cost-effectiveness, and extensive availability of validated kits and reagents. PCR's growing applications in detecting gene mutations with low allele frequencies further reinforce its clinical relevance. Complementary technologies—including next-generation sequencing (NGS), in situ hybridization (ISH), and immunohistochemistry (IHC)—continue to expand their share as precision requirements evolve.
By Geography: North America dominates the global landscape, led by the United States and Canada, where technological advancement and robust regulatory support from authorities such as the FDA accelerate product innovation and market entry. This regional strength reflects sustained investment in precision medicine infrastructure and a mature reimbursement ecosystem.
Request Sample Pages: https://www.marketsandmarkets.com/requestsampleNew.asp?id=155571681
THE OPPORTUNITY: RESHAPING DRUG DEVELOPMENT AND TIME-TO-MARKET
The co-development of companion diagnostics alongside therapeutic products represents a transformative opportunity for pharmaceutical enterprises. This integrated approach streamlines the drug development process, accelerates commercialization timelines, and enables the creation of safer, more effective medications with enhanced therapeutic efficacy—all while reducing development costs. Who stands to benefit? Therapeutic manufacturers seeking regulatory approval advantages, diagnostic companies pursuing strategic partnerships, and healthcare systems committed to evidence-based precision care.
A notable recent example illustrates this trajectory: In October 2022, F. Hoffmann-La Roche Ltd. received FDA approval for the first companion diagnostic to identify patients with HER2 low metastatic breast cancer, demonstrating the regulatory pathway's maturation and market receptivity to innovation.
CRITICAL MARKET HEADWINDS REQUIRE STRATEGIC ATTENTION
Despite growth prospects, the market faces two significant structural challenges that C-suite leaders must address:
Capital Requirements and Innovation Economics: The development, validation, and regulatory approval of biomarker-based diagnostics demand substantial capital investments. The pharmaceutical industry's phase III clinical trial failure rate—approximately 30% of drug candidates—creates compounding financial pressure on diagnostic manufacturers. Achieving successful phase III trials, followed by rigorous regulatory approval for in vitro diagnostic (IVD) products, requires considerable resources that disproportionately disadvantage smaller innovators and limit biomarker discovery velocity.
Workforce Capability Gaps: Companion diagnostic testing demands specialized technical expertise and proficiency in complex technologies. A critical shortage of adequately trained laboratory professionals—coupled with insufficient clinical awareness of emerging diagnostic advances—creates operational bottlenecks, particularly in emerging markets. Organizational resistance to transitioning from manual laboratory operations to automation and IT-based platforms further constrains adoption and market scaling.
MARKET LEADERSHIP AND KEY STAKEHOLDERS
The competitive landscape is anchored by global diagnostics and life sciences leaders:
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Agilent Technologies, Inc. (United States)
- QIAGEN (Netherlands)
- Thermo Fisher Scientific Inc. (United States)
- Abbott (United States)
These organizations are actively shaping market dynamics through innovation, regulatory approvals, and strategic positioning in high-growth therapeutic areas.
STRATEGIC IMPLICATIONS FOR DECISION-MAKERS
For pharmaceutical executives, the imperative is clear: integrate companion diagnostic development into therapeutic development strategies to accelerate approval timelines and improve market differentiation. For diagnostic manufacturers, the opportunity lies in addressing workforce capability gaps through training, automation, and technology partnerships. For healthcare system leaders, investment in precision diagnostic infrastructure directly correlates with improved patient outcomes and operational efficiency in value-based care models.
The companion diagnostics market's projected expansion to $13.6 billion by 2029 reflects not merely a financial milestone, but a fundamental reorientation of healthcare toward data-driven, personalized therapeutic approaches—a shift that will define competitive advantage across the global healthcare ecosystem.
Related Reports:
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email:Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com/Market-Reports/companion-diagnostics-market-155571681.html