The global rapid medical diagnostic kits market size is expected to reach USD 23.6 billion by 2027, expanding at a CAGR of 4.2%, according to a new report by Grand View Research, Inc. The growing prevalence of infections caused by multidrug-resistant bacteria has led to high demand for rapid diagnosis. Moreover, the outbreak of COVID-19, caused by a newly discovered coronavirus, has created a significant need for rapid diagnostic tests to manage and control the pandemic.
Some of the factors that have played a vital role in the rising prevalence of emerging diseases are genetic variation in hosts and pathogens, population pressure, and environmental change. Rapid detection of chronic diseases, especially infectious diseases, is necessary and should be accurate and affordable too. It helps minimize the need for multiple patient visits and facilitates timely treatment and control of infectious disease outbreaks such as COVID-19. In response to the pandemic, many manufacturers of diagnostic tests are focusing on the development of rapid and easy-to-use point of care COVID-19 diagnostic tests to facilitate testing outside of lab settings. For instance, on March 21, 2020, Cepheid, a subsidiary of Danaher Corporation, launched a novel COVID-19 diagnostic test namely Cepheid Xpert Xpress SARS-CoV-2 post Emergency Use Authorization (EUA) approval from the FDA.
Regulatory bodies such as WHO, CDC, and European Centre for Disease Prevention and Control, are engaged in active recording of data on incidence and prevalence of infectious diseases to control the disease systematically. These agencies work together with local governments to achieve overall control of the treatment and management of infectious diseases. For example, the Global Influenza Programme (GIP), by WHO, provides technical support, strategic guidance, and coordination activities concerning a zoonotic, pandemic, and seasonal influenzas.
Growing prevalence of infectious diseases including tuberculosis, malaria, HIV/AIDS, pneumonia, hepatitis, and influenza is one of the major factors propelling the market growth. According to WHO, around 58.0 million people were diagnosed and treated for tuberculosis between 2000 and 2018 across the globe. The WHO also stated that in 2018, over 228.0 million malaria cases occurred around the world, with over 93.0% cases being reported from Africa, 3.4% from Southeast Asia, and 2.1% from the Eastern Mediterranean region.
Request a Sample Copy of the Rapid Medical Diagnostic Kits Market Research Report @ https://www.grandviewresearch.com/industry-analysis/rapid-medical-diagnostic-kits-industry/request/rs1
Rapid Medical Diagnostic Kit Market Report Highlights
- Over-the-counter (OTC) kits dominated the market in 2019 and accounted for a share of 61.76% and is anticipated to maintain its dominance over the forecast period
- This is attributed to the fact that OTC rapid diagnostic tests allow frequent monitoring of disease/treatment method as these tests are easily available and can be performed without professional medical intervention
- The lateral flow segment dominated the market in 2019 and accounted for a share of 32.2%. The segment is expected to maintain this dominance over the forecast period
- Low development cost and easy manufacturing have led to the expansion of applications of lateral flow assays into areas where rapid testing is needed
Have Any Query? Ask Our Experts@ https://www.grandviewresearch.com/inquiry/192/ibb
Rapid Medical Diagnostic Kit Market Segmentation
Grand View Research has segmented the global rapid medical diagnostic kit market based on the product, technology, application, end-use, and region:
Rapid Medical Diagnostic Kit Product Outlook (Revenue, USD Million, 2016 - 2027)
- Over-The-Counter (OTC) Kits
- Professional Kits
Rapid Medical Diagnostic Kit Technology Outlook (Revenue, USD Million, 2016 - 2027)
- Lateral Flow
- Agglutination
- Solid Phase
- Other Technologies
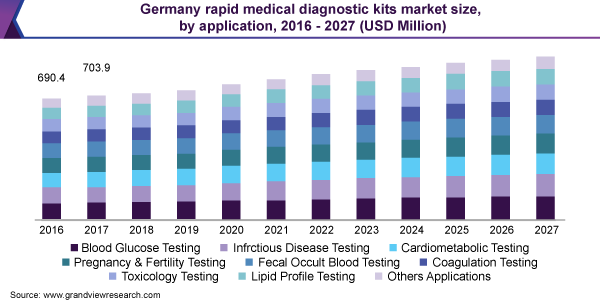
Rapid Medical Diagnostic Kit Application Outlook (Revenue, USD Million, 2016 - 2027)
- Blood Glucose Testing
- Infectious Disease Testing
- Cardiometabolic Testing
- Pregnancy and Fertility Testing
- Fecal Occult Blood Testing
- Coagulation Testing
- Toxicology Testing
- Lipid Profile Testing
- Other Applications
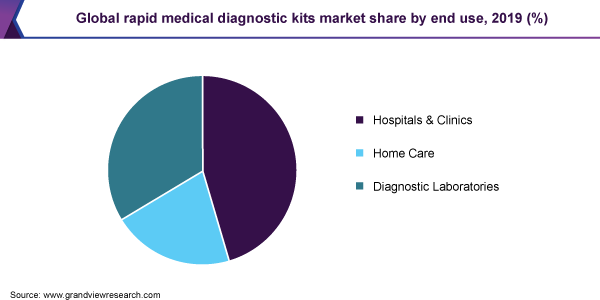
Rapid Medical Diagnostic Kit End-use Outlook (Revenue, USD Million, 2016 - 2027)
- Hospitals & Clinics
- Home Care
- Diagnostic Laboratories
Rapid Medical Diagnostic Kit Regional Outlook (Revenue, USD Million, 2016 - 2027)
- North America
- The U.S.
- Canada
- Europe
- Germany
- The U.K.
- France
- Italy
- Spain
- Russia
- Asia Pacific
- Japan
- China
- India
- South Korea
- Australia
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East Africa (MEA)
- South Africa
- Saudi Arabia
- UAE
List of Key Players of Rapid Medical Diagnostic Kit Market
- ACON Laboratories, Inc.
- Abbott Laboratories
- Artron Laboratories Inc.
- Alfa Scientific Designs, Inc.
- Becton, Dickinson and Company
- BTNX, Inc.
- bioMérieux SA
- Cardinal Health
- Bio-Rad Laboratories, Inc.
- Danaher Corporation
- Creative Diagnostics
- Meridian Bioscience, Inc.
- F. Hoffmann-La Roche AG
- McKesson Medical-Surgical, Inc
- Sight Diagnostics Ltd
- Trinity Biotech
- Zoetis
Browse Related Reports @
COVID-19 Diagnostics Market: https://www.grandviewresearch.com/industry-analysis/covid-19-diagnostics-market
Blood Transfusion Diagnostics Market: https://www.grandviewresearch.com/industry-analysis/blood-transfusion-diagnostics-market
About Grand View Research
Grand View Research provides syndicated as well as customized research reports and consulting services on 46 industries across 25 major countries worldwide. This U.S.-based market research and consulting company is registered in California and headquartered in San Francisco. Comprising over 425 analysts and consultants, the company adds 1200+ market research reports to its extensive database each year. Supported by an interactive market intelligence platform, the team at Grand View Research guides Fortune 500 companies and prominent academic institutes in comprehending the global and regional business environment and carefully identifying future opportunities.
Media Contact
Company Name: Grand View Research, Inc.
Contact Person: Sherry James, Corporate Sales Specialist - U.S.A.
Email:Send Email
Phone: 1-415-349-0058, Toll Free: 1-888-202-9519
Address:201, Spear Street, 1100
City: San Francisco
State: California
Country: United States
Website: https://www.grandviewresearch.com/industry-analysis/rapid-medical-diagnostic-kits-industry

